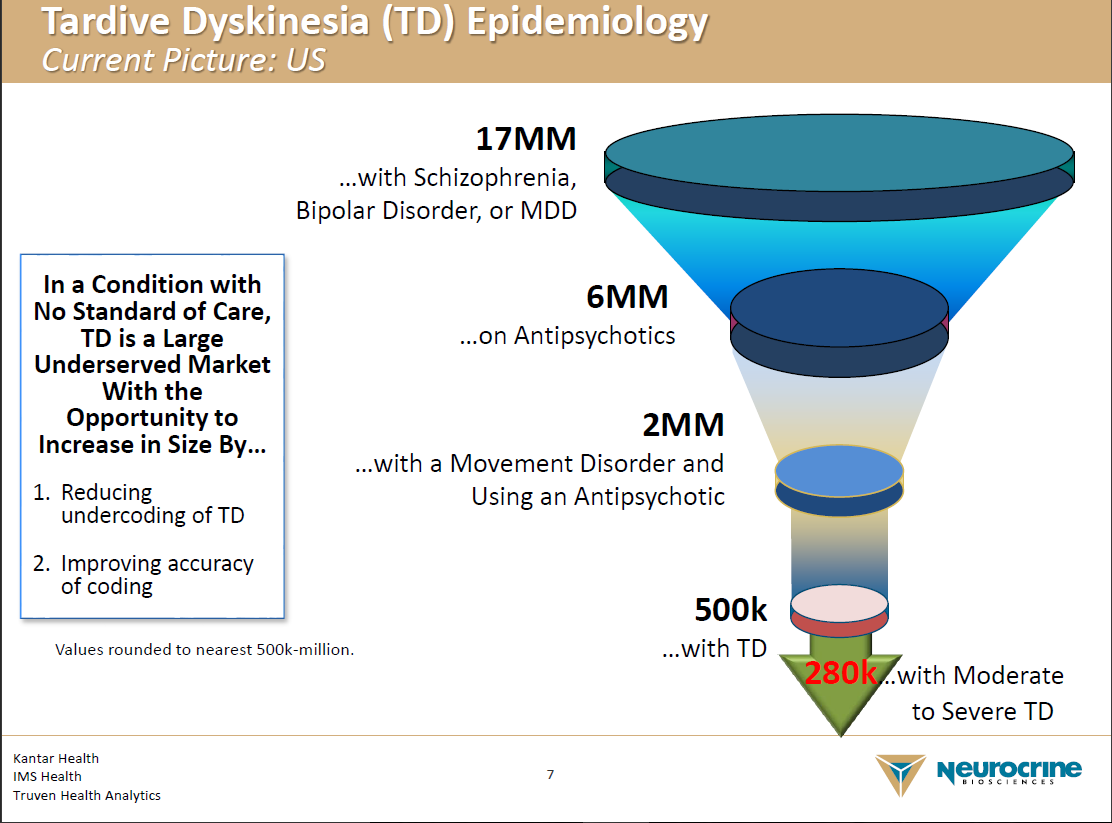
Austedo - Deutetrabenezina 12mg caixa com 60 comprimidos - TEVA - ÁgilMed - Medicamentos Especiais e Nutrição Clínica

Hungry for brand sales, Teva undercuts generics with new, $60K Huntington's med Austedo | Fierce Pharma
Austedo (deutetrabenazine) for the Treatment of Chorea Associated with Huntington's Disease - Clinical Trials Arena

Punit on Twitter: "#Austedo (deutetrabenazine) revenue & TRx analysis Q2-17- $1 M Q2-18- $44 M Q2-19- $96 M Q1-20- $122 M Now, Austedo approval in China may increase #API demand. Also,dose increases

Teva Announces FDA Approval of AUSTEDO™ (deutetrabenazine) Tablets for the Treatment of Chorea Associated with Huntington's Disease — Hereditary Neurological Disease Centre
Austedo (deutetrabenazine) for the Treatment of Chorea Associated with Huntington's Disease - Clinical Trials Arena